Osmosis Sucks
Why Cardiac Tamponade May Be Delayed
Reproduced From Shutterstock by Subscription
I was a fellow when I learned that osmosis sucks.
The year was 1976 before echocardiography was routine, and not long after the British pathologist, Robert Donald Teare, had described eight young people who had asymmetric cardiac hypertrophy of the ventricular septum. Seven of those individuals had died suddenly. Teare noted that their myocardium contained disorganized muscle bundles. This finding would later be recognized as the classical “myocardial disarray” of hypertrophic cardiomyopathy.
Teare is often credited with discovering the condition, but cardiologist Eugene Braunwald 1 credits a French physician named Henri Liouville who, in 1869, described a 75-year old woman whose left ventricular walls measured 3.5 to 4 cm in thickness. Liouville titled his publication Cardiac Sub-Aortic Stenosis. Several other reports also appeared before Teare’s, but Teare gets most of the credit, but I digress.
On a Friday in 1976, I was performing a cardiac catheterization on a patient whom I thought had aortic stenosis. I was using a catheter called the “long-tipped Lehman”, which the older fellows had nicknamed the “Lehman Spear”. The extended, tapered tip of the device was said to be soft and flexible, but we thought it became stiff, perhaps from sterilization. That made the catheter prone to penetrate the ventricle. At least some other fellows knew it penetrated ventricles but not me, a first-year cardiology fellow.
It is amazing the amount of independence we had as trainees. I was in the catheterization laboratory procedure room with the nurse, Kathy, and another cardiac fellow. I was doing the procedure. After I advanced the long tipped Lehman into the left ventricle, I was preparing to do the ventricular dye injection, using an osmotic dye (the only type of intravascular dye we had at the time), but I could not get blood to flow back from the catheter. I called my attending who, to my recollection, commented that I always was very careful, (the implication was too careful), and that I should go ahead and inject. I did and the pericardial space lit up with dye.
I was calm then, although I became quite anxious about it that night. I have had a similar experience of “delayed dread” when I used to fly to Nantucket, an island off the coast of southern Massachusetts, to put in pacemakers. I once took off from Nantucket, wound up in clouds without an instrument rating, and couldn’t go back to Nantucket because it was closed by fog! After I was vectored by Otis Airforce Base to Providence and finally landed, I couldn’t stop shaking. But again I digress.
I figured that the dye in the pericardial space would suck in additional fluid, so I asked Kathy to get me the pericardiocentesis needle. I watched as the patient’s heart rate went up and blood pressure went down. After about 10 minutes, when the patient’s systolic pressure got to about 100 mmHg, I tapped the pericardium. The fluid contained almost no blood.
That Saturday was my 10th high school reunion. I really wanted to go to see what happened to my high school girl friend. I did go briefly, but spent most of the weekend in the hospital watching my patient. She did fine. After looking at the left ventricular pressure tracings, it was clear that the patient had hypertrophic cardiomyopathy, which explained her “aortic stenosis” murmur and probably why the Lehman perforated her ventricle: the ventricle was small and hyperdynamic, so it pressed down hard on the catheter.
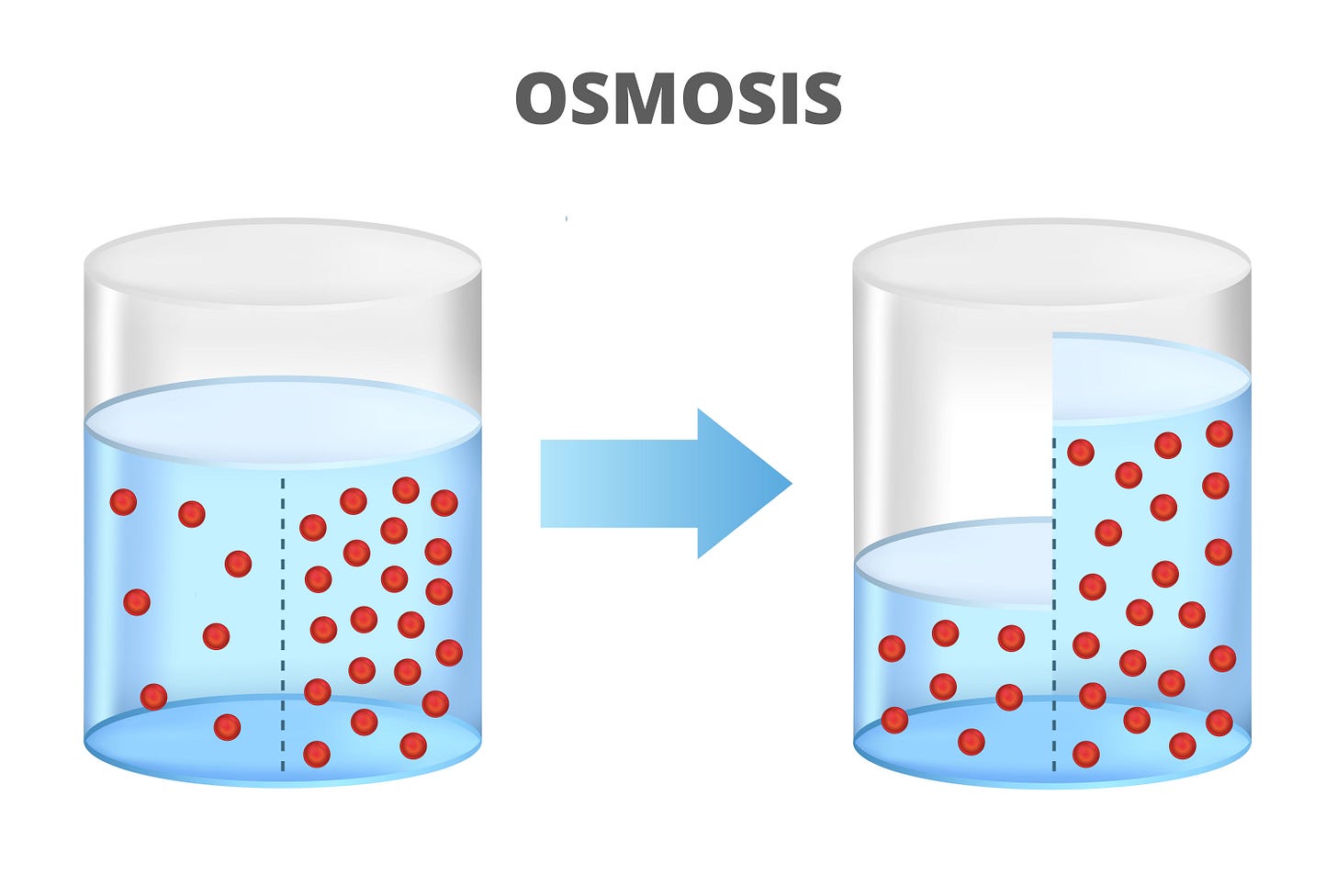
That was my first experience with pericardial suck, but I think I have seen other cases where osmosis lead to pericardial tamponade.
I first thought this process might occur when I took care of a patient who had fallen down the stairs and hit her chest on the wooden pineapple on the banister at the bottom of the stairs. She came in with classic tamponade two weeks later. The pericardial fluid was serosanguinous but not bloody. I wondered if the blow to her chest had produced a small amount of hemorrhage in the pericardial sack and that the RBCs fractured over time, producing an osmotic gradient leading to tamponade.
The concept is that small amounts of intact red blood cells (RBCs) in the pericardial space do not cause osmotic suck. But when the RBCs break down, they create an osmotic gradient.
I was not certain that blaming osmosis was correct, so before finishing this post I asked several experts. One of my concerns was that the potassium concentration released from the RBCs should create an osmotic pressure similar to the sodium pressure in plasma,. This would not create a gradient.
Sabet Hashim, MD, Head of Cardiac Surgery and Co-Physician in Chief of Hartford Healthcare’s Cardiovascular Institute, noted that hemoglobin itself also breaks down into its globins and heme, and the globins can breakdown further creating a higher osmotic gradient. Dr. Hashim told me that this was his explanation as to why some coronary bypass patients develop left-sided pleural effusions several weeks after harvesting the left internal mammary artery.
My good friend, Tim Lepore, MD, a general surgeon on Nantucket, noted that RBC hemolysis is thought to be the mechanism for expansion of thigh hematomas over time. This hemolysis concept was also once thought to explain subdural hematomas, but this process now appears considerably more complex. 2
Jon Hammond, MD, another cardiac surgeon at Hartford HealthCare, said, “I agree with the ‘osmotic suck’ idea. I have certainly seen post-op heart patients with retained blood in the pericardium develop tamponade a week or two later, although that theoretically could be attributed to some form of post cardiotomy syndrome as well.”
Ultimately I discussed this proposed mechanism for delayed tamponade with a pericardial disease expert, Dr. Allan Klein from the Cleveland Clinic. Dr. Klein sent me this quote from David Spodick’s book, The Pericardium: A Comprehensive Textbook, published first in 1997 and again in 2013 :
“ … persistent enlargement of pericardial contents may occur due to indrawing of fluid attracted by the osmotic effects of split proteins ( fibrinolysis) and other molecules that increase the number of intrapericardial contents.”
So, what are the Rules here?
1. Think of the possibility of cardiac tamponade even if several weeks have elapsed since a blow to the chest or a cardiac procedure that could have perforated the ventricle and introduced blood into the pericardium, such as a pacemaker placement or an ablation.
2. This delayed presentation may be due to other conditions such as post pericardiotomy syndrome, but some delayed presentations of tamponade are because some blood got into the pericardial space and …Osmosis Sucks.
References -
1. Braunwald E. Hypertrophic cardiomyopathy: The first century 1869-1969. Glob Cardiol Sci Pract 2012;2012:5.
2. Edlmann E, Giorgi-Coll S, Whitfield PC, Carpenter KLH, Hutchinson PJ. Pathophysiology of chronic subdural haematoma: inflammation, angiogenesis and implications for pharmacotherapy. J Neuroinflammation 2017;14:108.
#Pericardialtamponnade; #pericardium; #thighhematoma; #subduralhematoma; #AllanKleinMD; #Davidspodickmd